We apply our proprietary AP3 patient selection platform to develop targeted precision oncology medicines that we match to patients whose tumors are predicted to be sensitive to each specific medicine. Our clinical pipeline is initially focused on an advanced asset targeting the DNA Damage Response (DDR) and cell cycle regulation with demonstrated, durable single agent clinical activity in solid tumors but where genetics-based approaches have proven insufficient for response prediction. Likewise, our preclinical programs target critical nodes in cell cycle pathways where we believe genetics-based approaches are insufficient. Our OncoSignature patient selection method will be used to aim for patient responder enrichment to increase the likelihood of successful clinical development.

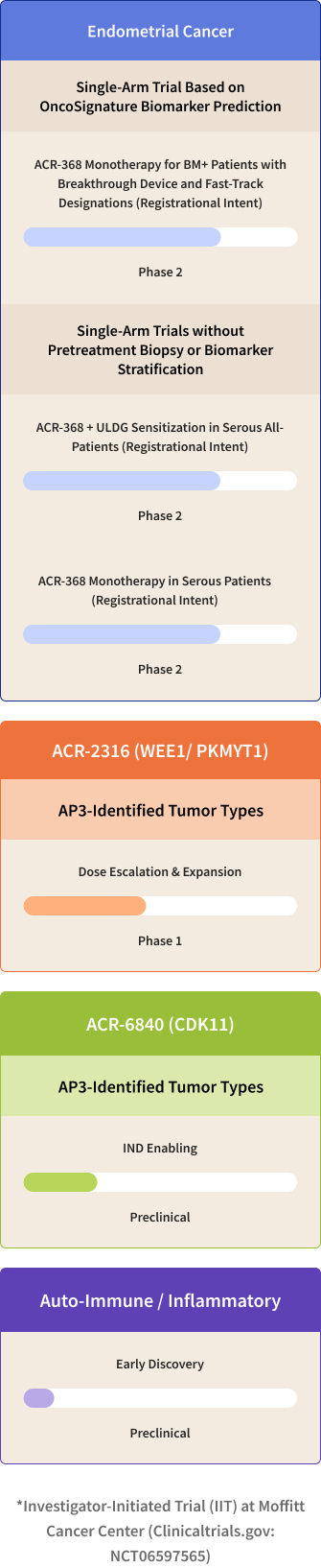
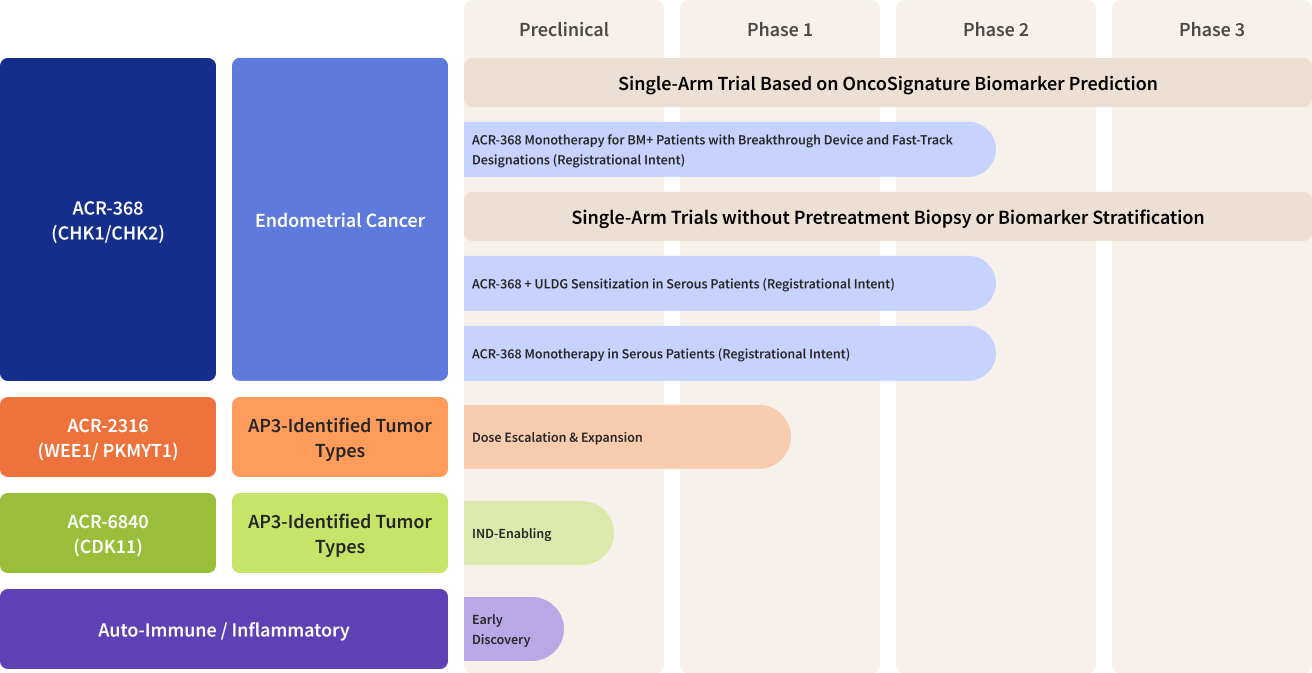
Acrivon Pipeline
Acrivon’s Lead Program ACR-368 (also known as Prexasertib, in-licensed from Lilly), is a clinically-advanced, potent selective inhibitor of the DNA Damage Response checkpoint kinases, CHK1 and CHK2. ACR-368 has demonstrated deep, durable single-agent anti-tumor clinical activity, including complete responses, in a proportion of patients with platinum-resistant ovarian cancer* and squamous cell cancer ‡. Using OncoSignature, we have identified endometrial cancer as a high unmet need solid tumor type predicted to be highly sensitive to the drug candidate. The company has received fast track designation from the Food and Drug Administration, or FDA, for the investigation of ACR-368 as monotherapy based on OncoSignature-predicted sensitivity in patients with endometrial cancer or platinum-resistant ovarian cancer. The FDA has granted Breakthrough Device designations for the ACR-368 OncoSignature assay for the identification of patients with endometrial cancer or for patients with ovarian cancer, who may benefit from ACR-368 treatment. Acrivon is also leveraging its proprietary AP3 precision medicine platform for developing its co-crystallography-driven, internally developed pipeline programs, consisting of its clinical candidate, ACR-2316, a selective, dual WEE1/PKMYT1 inhibitor and ACR-6840, a development candidate targeting CDK11. More information on our clinical trials can be found on clinicaltrials.gov, and information on our Expanded Access Policy can be found here.
*Ref: Lee et al, Lancet Oncology (2018); Konstantinopoulos et al, Gynec Onc (2022); ‡ Ref: Hong et al, CCR (2018)